The state is making good progress toward its goal of 100% clean electricity by 2040, generating over half of it with zero emissions last year, and with total emissions dropping 10% compared with 2022. On average, this sector has reduced its carbon footprint by 2 million metric tons of carbon dioxide (CO2) per year since 2018, putting us on track to complete this transition by 2032.
On its own, this sounds fantastic, but there are reasons to believe this progress may stall. Our increased reliance on electricity or electric-derived products like green hydrogen means demand for electricity is increasing faster than we are adding capacity. The cost of electricity has outpaced inflation by 20% since 2003, and there’s no sign of slowing. Furthermore, lack of existing infrastructure means current solar and wind projects are getting held up, slowing our progress and driving price increases.
Because of this, both Xcel and Minnesota Power are proposing new natural gas power plants to keep up with demand and make up for capacity lost from shuttered coal plants. Minnesota isn’t the only state feeling the pressure, either.
What if I told you there is a source of electricity that can entirely replace current fossil fuel plants without new transmission infrastructure? A source that, despite its reputation, has generated about one quarter of the state’s electricity needs for over 50 years safely, cheaply and with zero emissions? Believe it or not, nuclear power is that source.
However, therein lies another risk. As our two existing power plants exceed 50 years of age, we need to start thinking about what comes next. Their licenses expire in the next 10 years, and even if the licenses get renewed, the plants themselves will need replacing one day. The best time to start thinking about the future is now, and the best place to start is from the very beginning, so I’ll start by posing and answering a basic question.
What Even Is Nuclear Power?
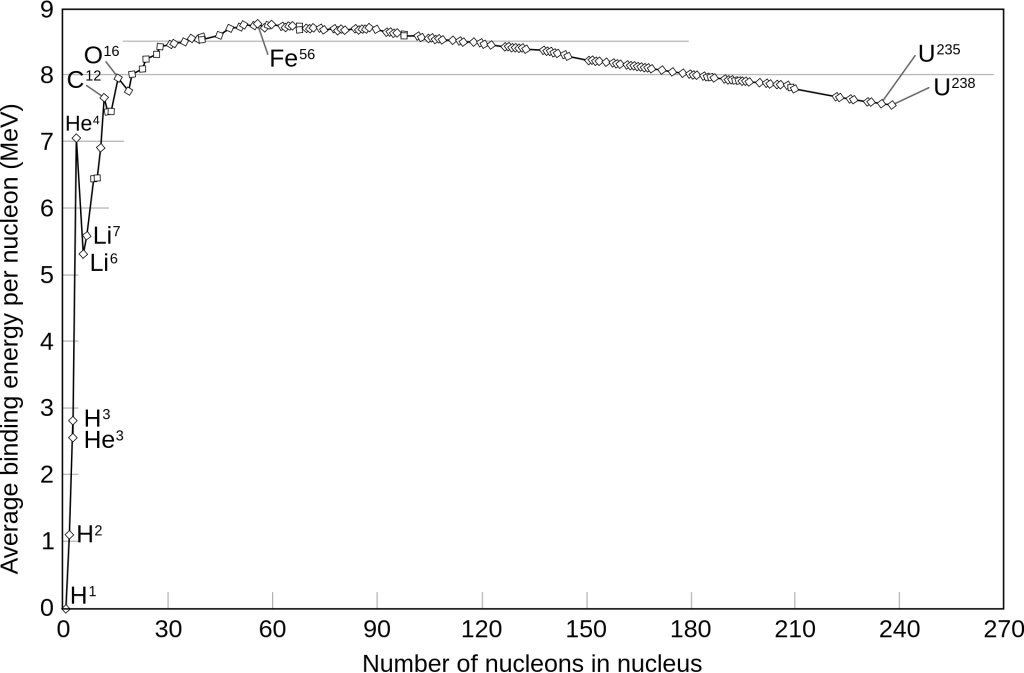
What we commonly call “nuclear power” is the process in which we generate electricity by the splitting — also called fission or decay — of atoms. All atoms have a minimum amount of energy required to split or fuse them, called the binding energy. This amount varies based on the number of protons and neutrons (called nucleons) in the nucleus of an atom. The force that binds subatomic particles to form nucleons (called the strong nuclear force) has residual effects that also attract those particles in other nucleons, holding them together at incredibly short distances.
These effects get stronger as the number of nucleons in an atom increases, but, as atoms get larger, the average distance between them increases, diminishing this force. Furthermore, a greater number of protons in the nucleus means a larger positive charge to repel them, and this doesn’t decay as much over the same distance as the strong force. Once atoms reach the size of iron, the electromagnetic force begins to overpower the strong nuclear force, resulting in a decrease in binding energy.
I’m going to stop here for a moment to introduce the Second Law of Thermodynamics, as it is the guiding principle from here all the way to the input shaft of the electric generator. Simply put, it states that things in a high energy state want to move to a lower one. While things can go from a lower energy state to a higher one, this process must also expend energy in such a way where the total amount of energy used over a full cycle (low to high to low again) is greater than zero. This is true of all things, not just individual atoms but chemical reactions, electricity, the weather — all the way up to gravitational forces between planets, stars, galaxies and so on. This is usually referred to as “potential energy” as it is energy that can be released when given the right impetus. This is also why there’s no such thing as free energy or perpetual motion.
Stability
Keep in mind: An increase in binding energy actually indicates a lower energy state. Again, all things like to be in a low energy state. The less energy something already has, the more it takes to move it out of that state, and binding energy is how we measure that amount of energy. If you’re struggling, try flipping the above graph upside down and thinking of it as a bowl. Replace the atom with a ball, and fission or fusion with moving left or right, respectively. A ball at the bottom of this curve needs a kick or throw to move in either direction, and binding energy is how we measure the strength of that kick or throw.
There’s more to stability than just the total number of nucleons. Several other factors, like the exact ratio of protons to neutrons and whether the number of each are odd or even also play a role. Elements are defined by the number of protons, and each can has a different number of neutrons, called isotopes. In elements larger than lead, the binding energy is low enough that all isotopes can spontaneously* decay. This is the difference between stable and unstable elements. All stable elements also have unstable isotopes, and many have multiple stable ones as well.
* Doesn’t spontaneity violate Newton’s First Law?
Ok, fine, it’s not actually spontaneous, but I’m trying to keep things as simple as possible, which means drawing a line somewhere. The reasons behind “spontaneous” fission are deeply entwined in quantum mechanics, so even if I fully understood them (which I don’t), I would need a whole other article to explain what is ultimately a minor detail. “Spontaneous Fission” is commonly accepted shorthand, so we’ll stick with it.
Going back to the ball analogy, a ball on a steeper slope can overcome friction and roll to the bottom, while ones on a shallower slope might stick and stay put.
Although almost all atoms can be split with enough energy, smaller elements usually do not give out more energy than they require to split since the amount binding them is so low and they have few nucleons. Likewise, it takes massive amounts of energy to fuse large elements together to form giant ones, due to the large electromagnetic forces trying to push them apart. However, something counterintuitive happens when splitting large atoms: they actually emit more energy than it takes to break them!
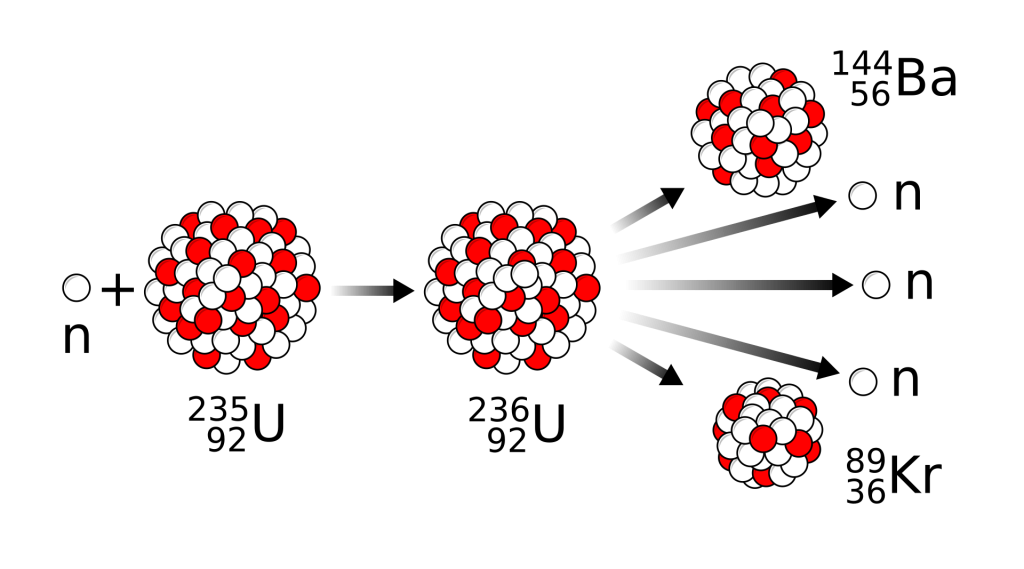
E =mc2
In order to understand why, we must consult that most famous of equations, or rather, a slight variation: E = Δmc2. In mathematics, a Δ (Delta) just means a change or difference. For example, if you add up the masses of 89KR, 144BA and the three neutrons in the decay chain above, the number you’ll get is actually slightly smaller than the mass of 236U. This difference is the value of Δm, and is what’s known as the mass defect. In essence, this extra mass is how and where the binding energy is stored. Likewise, after the atom splits, this mass is turned into a quantity of energy proportional to the speed of light (c) squared.
Some of this energy is transferred into the fission products in the form of kinetic energy (i.e., movement), while the rest is released as a photon — the carrier of all electromagnetic radiation, like light or radio waves. These particles then go and bump into other nearby particles, resulting in an increase in temperature in the surroundings, as thermal energy is just mean kinetic energy. This is why you can cook a chicken by slapping it.
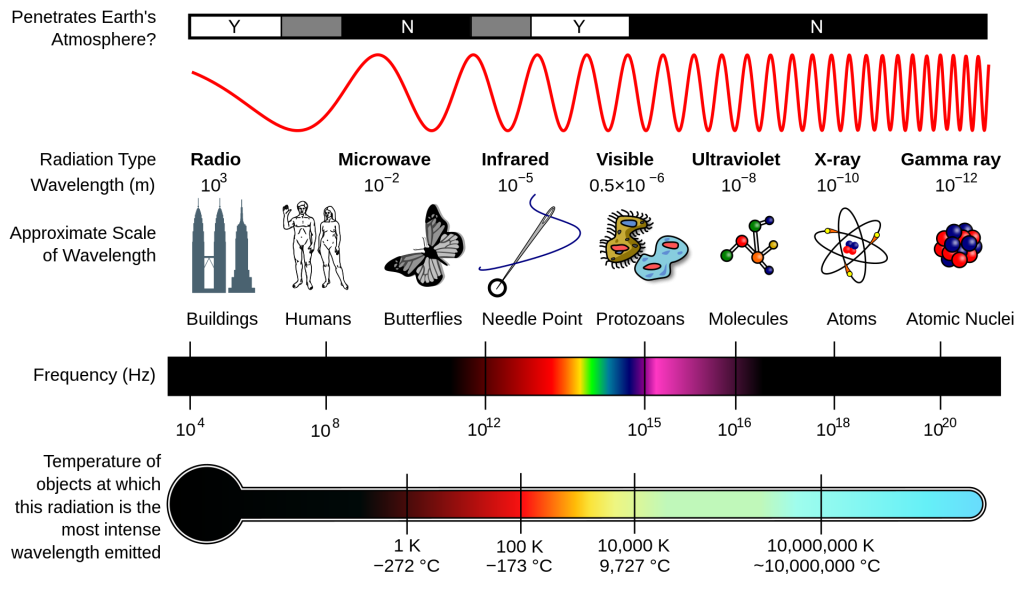
Side note: The inverse of all of this is also true. Small elements need a large amount of kinetic energy in order to overcome electromagnetic repulsion and fuse, meaning the resultant product will have a smaller mass than the sum of the two starting products, and this mass defect is the energy released. While research into using fusion for electricity generation is coming along, I’ll be sticking to fission reactors, as those are currently in use and the bulk of what’s being built in the near future.
Spontaneous fission is a process that typically happens so slowly we can actually see individual events. You’ve probably heard the term “half life” before. This is the amount of time it takes for half of a radioactive substance to decay, and tends to be in the range of millions to billions of years for naturally occurring radioactive elements. As a Boy Scout, I helped build a cloud chamber as part of my nuclear science merit badge to witness these events. While we used it to look at cosmic radiation, any radioactive sample works.

This actually presents a problem: Under normal conditions, many radioactive substances emit so little energy we can’t do anything with it; in fact, even with proper equipment, it can be hard to detect!
This doesn’t mean they’re lacking in energy. Some of you might remember when I compared the energy density (how much energy you can store in a given volume or mass) of various substances in my trolleybus article:
| Energy Storage | Specific Energy (MJ/kg) |
| Batteries | |
| Lithium nickel cobalt aluminum oxides | 0.79 |
| Lithium nickel manganese cobalt oxide | 0.74 |
| Lithium cobalt oxide | 0.70 |
| Lithium iron phosphate | 0.32-0.58 |
| Lithium titanate | 0.22-0.40 |
| Lead-Acid | 0.11-0.14 |
| Fuel-based solutions | |
| Hydrogen | 120-142 |
| Gasoline (pure) | 46.4 |
| Gasoline (E10) | 43.5 |
| “Gasoline” (E85) | 33.1 |
| Diesel | 45.6 |
| Natural gas | 53.6 |
I’ve adjusted the units slightly, so that it matches this handy graph that XKCD made with Uranium added.
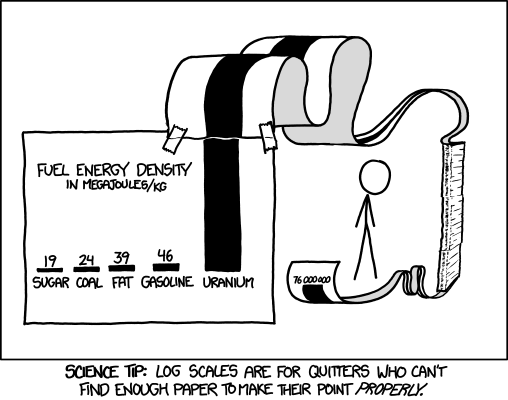
To put it another way: You would need to burn an Olympic-sized swimming pool full of gasoline to release the same amount of energy that’s in 1 kilogram of Uranium — about the size of a baseball. The problem is how we extract it. We already know how to burn stuff, we’ve been doing it for quite a long time now. One of the main reasons historically to burn stuff is to boil water.
On to Bigger and Better Things
We can use nuclear energy for that, too, but in order to usefully extract the energy from uranium, we’re going to have to speed up the process. You might not want to hold it, but if you did, that lump of uranium wouldn’t feel all that warm. With some clever engineering, we can get these fission events to trigger other ones, and those to trigger even more until suddenly we’re generating thousands of megawatts of power! This is also where the name of the device — a reactor — comes from, as reaction is a general term for turning one substance into another.
What kind of reactors should we build? Or, perhaps more importantly, what are the different kinds of reactors, and how do they work? How do we make sure these chain reactions don’t get out of control? What are the costs and benefits of each? How do they compare to other types of power generation? These are all questions that will need to wait for subsequent parts. In part 2, I’ll explain the different ways we go from fission to electricity, as well as the costs and benefits of each design.